lordjeff said:
The Sun is the prime driver of climate, not a simple gas that occupies 0.038% of the atmosphere.
This is a typical argument from incredulity on how CO₂ at concentrations of around 400 ppm can have a effect on temperature.
The answer lies in a combination of quantum mechanics and
statistical mechanics.
As discussed
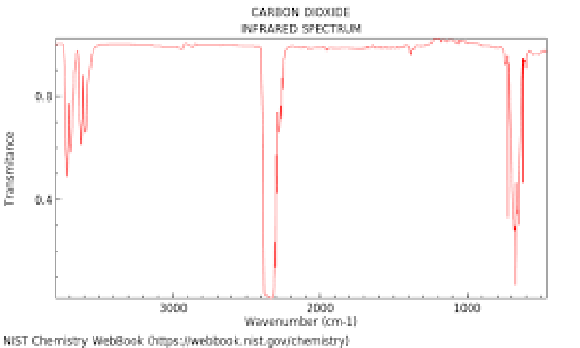
here CO₂ absorbs IR at 15μm wavelength.
When this occurs CO₂ molecules go into an excited transition state before returning to the ground (unexcited) state by emitting IR.
This IR radiation when directed to the surface and heats it is the mechanism for the greenhouse effect.
A secondary mechanism for heating is the collision of excited CO₂ molecules in the atmosphere.
According to the energy time version of
Heisenberg’s uncertainty principle;
ΔEΔt ≈ h/2 where h is Planck’s constant.
ΔE is the uncertainty in the energy measurement obtained by measuring the
FWHM (full width half maximum) of the 15μm peak which is the right hand side peak of the CO₂ IR absorption spectrum.
The spectrum is obtained at low CO₂ pressure and temperature to minimize peak broadening affects due to molecular collisions.
The excited transition state is found to be Δt = 6μs or a radiative decay rate of
R₁ = 1/Δt = 1.7 x 10⁵ s⁻¹.
Using statistical mechanics at a temperature of 288K (= 15⁰C) which is the blackbody temperature at which IR is radiated around 15μm and at 1 atm pressure (at sea level), with a CO₂ concentration of 400 ppm, a CO₂ molecule travels an average time of around 0.0001μs before colliding with dominant air molecules such as N₂ or O₂.
This is equivalent to a collision rate of R₂ = 28 x 10⁵ s⁻¹.
The probability an excited CO₂ molecule will return to the ground state without colliding into an air molecule is R₁/(R₁ +R₂) ≈ 0.06.
When an excited CO₂ molecule collides into air molecules, part of the excitation energy is converted into extra kinetic energy for the air molecules.
Since the average kinetic energy of the air molecules is now higher, the temperature of the atmosphere also increases.
Since 94% of collisions between a CO₂ molecule and air molecules occur with the CO₂ molecule in the excited state, a single excited CO₂ molecule engages in about 0.96 x 28 x 10⁵ ≈ 2,700,000 collisions every second.
Hence even at concentrations of 400 ppm, CO₂ affects temperature.
In fact if there was no CO₂ the average temperatures would drop around 7⁰C.