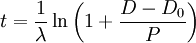Astridhere
Well-Known Member
- Jul 30, 2011
- 1,240
- 43
- Faith
- Christian
- Marital Status
- Married
I am quite familiar with the "creationist" literature. The British Journal Nature is probably the top science journal on the planet. New Scientist is a magazine about science. It is not a scientific journal.
This thread is not about the age of the earth, it is about dating methods that show the earth is older than 6,000 years.
You will have to find a dating method that is credible before you can have that conversation with anyone. The earth is not a closed system and only the hopefull would assume that no ground water, heat or anything else has interfered with your sample.
Quite frankly it is your face that is being slapped because you have cited a paper that does not support what you claim it does. That is why I keep asking you to give your critique of it. I know you have only read the abstract, not the paper itself. You have absolutely no idea what the U238/U235 ratio is or what it means. Here's a clue, that ratio has nothing to do with decay rates. And here's a fact you are completely ignorant of because you haven't read the actual paper. The ratio correction is only a difference of 700 k years at 4.5 Ga. The age of the earth is currently estimated at 4.54 Ga. +/- 1%. That 700 k is less than the current reported margin of error.
Yeah, like I said, you haven't a clue what that 137.88 ratio means. You also don't know that that ratio reported in your link, 30 March 2012 in Science, has been in use several years already any way. Brennecka. et al, published that ratio in 2009.
Oh really? The British Geological Survey states the following in a March news release concerning the paper we are discussing.
"A major effect of this work will be to decrease all previous uranium-lead (U-Pb) age determinations, by up to 700,000 years for samples that are about 4.5 billion years old the age of the Earth. In particular, the new 238U/235U ratio will allow geologists to place more accurate limits on the exact timing of a broad range of geological processes, from the initial formation of our planet, continents and economic mineral deposits, to past evolutionary events and climate change."
In Uranium-Lead dating, scientists measure the relative amount of U238 and U235 isotopes present in a sample, and then calculate the age. Too bad they keep getting it wrong. Do you reckon they got it right this time, finally?
You may minimize all you wish. Don't forget that this is the 4th change where each time you would have thought you've got it now. Well you didn't, and the sm146 half life is still changing.
You really should try reading the paper dear, instead of trying to give a spin to a subject of which you are completely ignorant.
Like I said above, "a field in which you are completely ignorant of". There are two kinds of isotopes, stable and unstable. Stable isotopes do not decay. Unstable isotopes decay at a specific constant rate. Unstable does not mean a varying rate.
No, but you do.
I would suppose the polite thing would be to say that you are uninformed and mistaken. But in reality....well, we all know, the roll you are playing, and it is more than obvious.
You can think what you like RickG. I suggest you were not aware of the research I posted.
The point still remains. You have 2 methods to date the earth and both of them have fallen into disrepute with one of them having the half life re-evaluated 4 times over 60 years.
The 'better' method did not confirm the previous sm146 dates but altered them every time. True scientists call this date different, despite the fact that it may be small in comparison to 4 billion years. True scientists also don't like this discepency, because a discrepency in dating is what it is, whether or not you care to admit it.
The same requirements are necessary as with all radiometric dating. You need a closed system you do not know you have. Once you get past this assumption, with Uranium used for dating, you then again assume that there was no lead present to begin with. The same goes for argon.
Your scientists can enter different insertion values as they please when guestimating. They sometimes assume 50% lead and 50% uranium initial quantities, or others assume 50% lead / 50% uranium initial quantities in addition to a loss of 50% uranium per half life with the loss of uranium through leaching with weak acids, another hopefull guestimate.
There appears to be many assumptions that you are able to adjust to give an intended ball park age that aligns somewhat with your preconcieved assumptions of what the age should be.
Despite all your credentials the above is the true state of the matter. Everchanging half lives only add to the assumptions that you have had to assume already.
Is it not so that in actual fact the result wanted will inform the insertion value to get the required date?
You guys have to have something and this is the best you've got. It seriously is no better that the 'any possible scenario' that litters all evolutionary literature.
In actual fact scientists have no idea what the initial content of the rock was and can only hope and guestimate at best. It is unlikely that any rock remains in a closed system for a million years let alone 4 billion. Now they do not even have reliable half life dates either, it would appear. Now you can have as many conversations you want and a boot load of credentials and still those facts will never go away.
Radiometric dating sadly tells us nothing for either side of the debate.
That is the state of your dating methods. Your dating methods demonstrate that some people like to play with assumptions. However it is another assumption to suggest they are credible or accurate.
Upvote
0