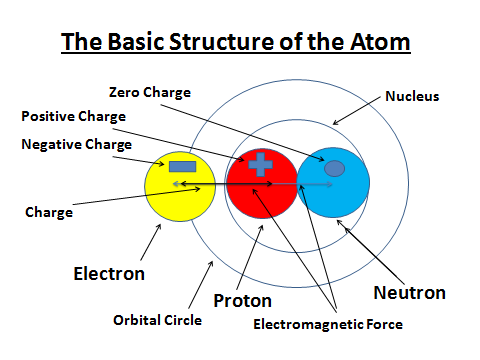
First of all, the accompanying diagram below does not represent Hydrogen Atom. Hydrogen does not have Neutron. But, rather it is the elements of the Periodic Table reduced in their 1:1:1 Neutron, Proton, and Electron ratios for simple analysis. The contents of the Atomic Structure diagram have what they come to represent in the Life of God.
The Atomic Elements
The Atom is made of the Nucleus which consists of Protons and Neutrons. The Protons and Neutrons are held together in the Nucleus by a strong nuclear force. The Electrons orbit around the Nucleus.
The Charges
The Proton and Electron are oppositely charged. The Proton has a Positive electric Charge, the Electron has a Negative electric Charge, and the Neutron has no electric Charge.
Equality of Charge in Particles
The Atom generally has a neutral overall Charge as a result of it having the same number of Electrons as Protons. It shows that the Nucleus of an Atom is always positively charged.
Equality of Mass in Particles
The Proton has the same mass as the Neutron, Electron’s mass is so small that this can usually be taken as zero.
The Atomic Number of the Atom
The Atomic Number (also called the Proton Number) is the number of Protons in an Atom. The Mass number (also called the Nucleon Number) is the total Number of Protons and Neutrons in an Atom.
Summary of the Atomic Structure
The statements of information about the Chemistry of the Atom Structure show the summary in this way: starting with the proton, it means that it has 1 on Mass and +1 on Charge; the neutron has 1 on Mass and zero (0) on Charge; and the Electron, zero (0) or 0.0005 on Charge. Such can be represented in tabular form in this way:
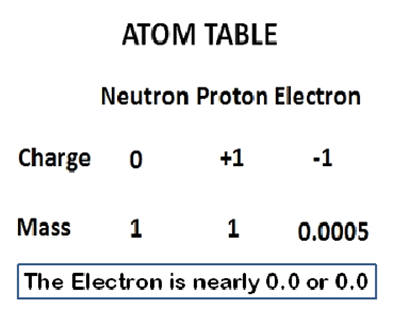
In the next post, we will be marrying the Divine Life Table derived as the summary of the Life of God the Holy Trinity in their internal Divinity and external Mission Activity and then with the Atom Table derived as the summary of the Atomic Structure. By that, we will be doing the decoding; which is the testing for the same or similar sentence structure, wording, and meaning.
The Atomic Elements
The Atom is made of the Nucleus which consists of Protons and Neutrons. The Protons and Neutrons are held together in the Nucleus by a strong nuclear force. The Electrons orbit around the Nucleus.
The Charges
The Proton and Electron are oppositely charged. The Proton has a Positive electric Charge, the Electron has a Negative electric Charge, and the Neutron has no electric Charge.
Equality of Charge in Particles
The Atom generally has a neutral overall Charge as a result of it having the same number of Electrons as Protons. It shows that the Nucleus of an Atom is always positively charged.
Equality of Mass in Particles
The Proton has the same mass as the Neutron, Electron’s mass is so small that this can usually be taken as zero.
The Atomic Number of the Atom
The Atomic Number (also called the Proton Number) is the number of Protons in an Atom. The Mass number (also called the Nucleon Number) is the total Number of Protons and Neutrons in an Atom.
Summary of the Atomic Structure
The statements of information about the Chemistry of the Atom Structure show the summary in this way: starting with the proton, it means that it has 1 on Mass and +1 on Charge; the neutron has 1 on Mass and zero (0) on Charge; and the Electron, zero (0) or 0.0005 on Charge. Such can be represented in tabular form in this way:
In the next post, we will be marrying the Divine Life Table derived as the summary of the Life of God the Holy Trinity in their internal Divinity and external Mission Activity and then with the Atom Table derived as the summary of the Atomic Structure. By that, we will be doing the decoding; which is the testing for the same or similar sentence structure, wording, and meaning.
