http://naturalselection.0catch.com/Files/ancientice.html#Oxygen and Other Isotopes
Why don't we study how isotopes actually behave in ice.
"The short-term peaks of d18O in the ice sheets have been ascribed to annual summer/winter layering of snow formed at higher and lower air temperatures. These peaks have been used for dating the glacier ice, assuming that the sample increments of ice cores represent the original mean isotopic composition of precipitation, and that the increments are in a steady-state closed system.
Experimental evidence, however, suggests that this assumption is not valid, because of dramatic metamorphosis of snow and ice in the ice sheets as a result of changing temperature and pressure. At very cold Antarctic sites, the temperature gradients were found to reach 500°C/m, because of subsurface absorption of Sun radiation. Radiational subsurface melting is common in Antarctica at locations with summer temperatures below -20°C, leading to formation of ponds of liquid water, at a depth of about 1 m below the surface. Other mechanisms are responsible for the existence of liquid water deep in the cold Antarctic ice, which leads to the presence of vast sub-sheet lakes of liquid water, covering an area of about 8,000 square kilometers in inland eastern Antarctica and near Vostok Station, at near basal temperatures of -4 to -26.2°C. The sub-surface recrystallization, sublimation, and formation of liquid water and vapor disturb the original isotopic composition of snow and ice. . .
Important isotopic changes were found experimentally in firn (partially compacted granular snow that forms the glacier surface) exposed to even 10 times lower thermal gradients. Such changes, which may occur several times a year, reflecting sunny and overcast periods, would lead to false age estimates of ice. It is not possible to synchronize the events in the Northern and Southern Hemispheres, such as, for example, CO2 concentrations in Antarctic and Greenland ice. This is, in part the result of ascribing short-term stable isotope peaks of hydrogen and oxygen to annual summer/winter layering of ice. and using them for dating. . .
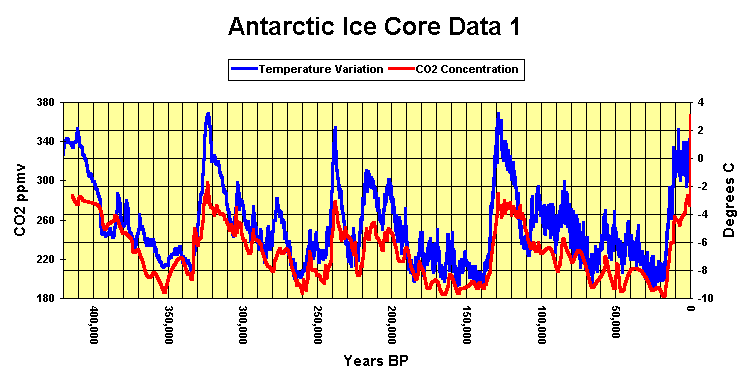
In the air from firn and ice at Summit, Greenland, deposited during the past ~200 years, the CO2 concentration ranged from 243.3 ppmv to 641.4 ppmv. Such a wide range reflects artifacts caused by sampling or natural processes in the ice sheet, rather than the variations of CO2 concentration in the atmosphere. Similar or greater range was observed in other studies of greenhouse gases in polar ice."
http://www.warwickhughes.com/icecore/
As for annual layers, some of us are quite aware as to how they are determined:
Of course those who support isotope dating will ignore that Glacier Girl found in 260 feet of ice 50 years later was found beneath hundreds of so called annual layering rings which they of course claim supports their isotope data.
"“After the snow deposition on polar ice sheets, not all the chemical species preserve the original concentration values in the ice. In order to obtain reliable past-environmental information by firn and ice cores, it is important to understand how post-depositional effects can alter the chemical composition of the ice.These effects can happen both in the most superficial layers and in the deep ice. In the snow surface, post-depositional effects are mainly due to re-emission in the atmosphere and we show here that chloride, nitrate, methane-sulphonic acid (MSA) and H2O2 [hydrogen peroxide] are greatly affected by this process; moreover, we show how the mean annual snow accumulation rate influences the re-emission extent. In the deep ice, post-depositional effects are mainly due to movement of acidic species and it is interesting to note the behavior of some substances (e.g. chloride and nitrate) in acidic (high concentrations of volcanic acid gases) and alkaline (high dust content) ice layers . . . We failed to identify any consistent relationship between dust concentration or size distribution, and ionic chemistry or snowpack stratigraphy.”
http://www.researchgate.net/publica...the_Penny_Ice_Cap_Baffin_Island_Arctic_Canada
http://www.detectingdesign.com/images/AncientIce/Greenland_Melt.jpg
http://www.detectingdesign.com/images/AncientIce/Portage Glacier Melt.jpg
http://www.detectingdesign.com/images/AncientIce/Pasterze Glacier Melt.jpg
Of course this is extensively used by those who promote global warming - but always excluded by those who support layering data and dating. So we can reasonably presume, can we not, that during the Middle Holocene where scientists believe the global temperatures were 2 to 5 degrees C warmer - that even more melting would have occurred?