New experiments done this year for the RATE project1 strongly support a young earth. This article updates results announced in an ICR
Impact article last year2 and documented at a technical conference last summer.3 Our experiments measured how rapidly nuclear-decay-
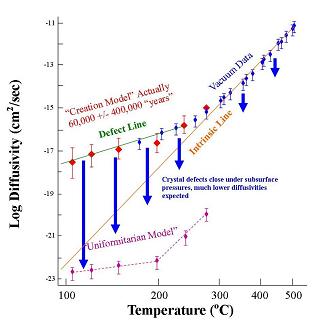
generated Helium escapes from tiny radio-active crystals in granite-like rock. The new data extend into a critical range of temperatures, and they resoundingly confirm a num-erical prediction we published several years before the experiments.4 The Helium loss rate is so high that almost all of it would have escaped during the alleged 1.5 billion year uniformitarian5 age of the rock, and there would be very little Helium in the crystals today. But the crystals in granitic rock presently contain a very large amount of Helium, and the new experiments support an age of only
6000 years. Thus these data are powerful evidence against the long ages of uniformitarianism and for a recent creation consistent with Scripture. Here are some details:
Radioactive crystals make and lose Helium
These radioactive crystals, called
zircons, are common in granitic rock. As a zircon crystal grows in cooling magma, it incorporates Uranium and Thorium atoms from the magma into its crystal lattice. After a zircon is fully formed and the magma cools some more, a crystal of black mica called
biotite forms around it. Other minerals, such as quartz and feldspar, form adjacent to the biotite.
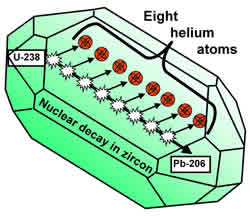
The Uranium and Thorium atoms inside a zircon decay through a series of intermediate elements to eventually become atoms of Lead. Many of the inter-mediate nuclei emit
alpha particles, which are nuclei of Helium atoms. For zircons of the sizes we are considering, most of the fast-moving alpha particles slow to a stop within the zircon. Then they gather two electrons apiece from the surrounding crystal and become Helium atoms. Thus a Uranium 238 atom produces eight Helium atoms as it becomes a Lead 206 atom. (See diagram.)
Helium atoms are lightweight, fast-moving, and do not form chemical bonds with other atoms. They move rapidly between the atoms of a material and spread themselves as far apart as possible. This process of
diffusion, theoretically well-understood for over a century, makes Helium leak rapidly out of most materials.
Natural zircons still contain much Helium
In 1974, in the Jemez Mountains of northern New Mexico, geoscientists from Los Alamos National Laboratory drilled a borehole several miles deep into the hot, dry granitic rock to determine how suitable it would be as a geothermal energy source. They ground up samples from the rock cores, extracted the zircons, and measured the amount of Uranium, Thorium, and Lead in the crystals. From those data they calculated that 1.5 billion years worth of nuclear decay had taken place in the zircons,6 making the usual uniformitarian assumption that decay rates have always been constant.7
Then they sent core samples from the same borehole to Oak Ridge National Laboratory for analysis. At Oak Ridge, Robert Gentry (a well-known creationist) and his colleagues extracted the zircons, selected crystals between 50 and 75 µm (0.002 to 0.003 inches) long, and measured the total amount of Helium in them. They used the Los Alamos Uranium-Lead data to calculate the total amount of Helium the decay had produced in the zircons. Comparing the two values gave the percentage of Helium still retained in the zircons, which they published in 1982.8
Their results were remarkable. Up to 58 percent of the nuclear-decay-generated Helium had not diffused out of the zircons. The percentages decreased with increasing depth and temperature in the borehole. That confirms diffusion had been happening, because the rate of diffusion in any material increases strongly with temperature. Also, the smaller the crystal, the less Helium should be retained. These zircons were both tiny and hot, yet they had retained huge amounts of Helium!