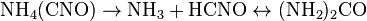Honestly, what is being argued here? What is True_Blue trying to say?
He keeps on as if there's some "ineffible" quality about life such that he has declared some possible reactions "off limits" because they are a violation of the Second Law of Thermodynamics.
How many times must someone post this equation? I dunno, but I'll do it yet again in hopes that True_Blue will actually address the science rather than throw out more meaningless rhetoric:
DG = DH - TDS
So, True_Blue, what about it? You are obsessed on the linearity or non-linearlity of DS, who cares about that? Please tell me how biochemical systems cannot contain any negative DS terms? Because we know that some biochemical processes (like the attachment of a small molecule to a protein, for instance) result in a loss of rotational degrees of freedom and therefore a loss of entropy, but the key is that the larger overall system gains in entropy (ie water displaced from the surface of the protein).
You seem to be oversimplifying the system again. As if Entropy is somehow totally antithetical to natural life to the point that life cannot exist without some supernatural intervention. Is that your stance?
Because it is demonstrably wrong to assume all entropy is always and ever increasing at every point in every system.
I hope ultimately you will address details of your "hypothesis", but I also realize this is very hard for you, owing to your not knowing much chemistry.
But that is precisely why you need to be careful in telling chemists how they are mistaken.
Please, start with one simple aspect of biochemistry that you can provide quantities for that proves that life is impossible without supernatural intervention.
I dearly would love to better understand what you are on about here and how it provides you with some proof of God's intervention.
I suspect what you are claiming to be further "proof" of God is actually just a failure on your part to take into account all the balances of entropy.
(in other words if you think one reaction that results in a decrease in entropy is ipso facto proof that God must be in command, I would imagine a scientist could come along and point out that indeed this reaction does show a DECREASE in entropy but only because a linked, and perfectly natural process is showing a concommittant increase in entropy. You have merely opted for a "God of the Gaps" hypothesis in a place where most of the gaps are filled already. This is why it would really help if you would learn enough chemistry so you could more effectively express your hypothesis.)
Interestingly, Thaumaturgy, several months ago I went out looking for scientists and scientific articles on the thermodynamics of photosynthesis in order to find the answer to a very specific technical question. After weeks of searching, I was unable to find what I was looking for. Most scientists specialize in very narrow areas, which works well most of the time, and an interdisciplinary approach to science is rare.
Anyways, you're looking at thermodynamics through a soda straw. Life exists in the presence of entropy because once life comes into being, it's mechanical processes are sustained by the influx of far more exogenous energy (90-99.9%) from the outside than the work that the life form generates. But that's just one narrow aspect of thermodynamics. The other aspects relate to the organization construct of the life form itself, similar to information entropy and the other forms of entropy I linked to earlier. Information entropy is one of many facets of a larger law at work. Thus, you can keep pouring gasoline into a car engine to allow the engine to operate despite the large amount of heat lost to entropy, but over time, the car engine itself will inevitably break down because the orderliness of the information contained in the engine will lose cohesion.
"But, at a more philosophical level, connections can be made between thermodynamic and informational entropy, although it took many years in the development of the theories of statistical mechanics and information theory to make the relationship fully apparent. In fact, in the view of Jaynes (1957), thermodynamics should be seen as an application of Shannon's information theory: the thermodynamic entropy is interpreted as being an estimate of the amount of further Shannon information needed to define the detailed microscopic state of the system, that remains uncommunicated by a description solely in terms of the macroscopic variables of classical thermodynamics."
http://en.wikipedia.org/wiki/Information_entropy
My intuition that entropy is nonlinear seems to have been proven out in Shannon entropy, or information entropy. I'm reasonably sure that all forms of entropy, once properly described, will be shown to be non-linear.
To answer your question about biochemistry, consider the coin toss chart on this link.
http://en.wikipedia.org/wiki/Information_entropy. When molecules react to form the 4 base units of DNA, the 4 units must organize themselves in specific ways to create a life form with functionality. Think of it as a four-sided coin. If you assume they have a 75% chance of creating a pattern condusive to organized life every time each base unit comes together, take .75 to the power of the minimum number of base pairs needed to make a functional life form. The probability of success follows the profile of the chart, and crashes rapidly and non-linearly to zero.
I recall many years ago seeing an interesting chart showing fitness over time for a healthy adult who eats right, exercises, and doesn't smoke. They are able to maintain a high level of fitness that decayed very slowly until the reached their early 80s, then their bodies would suddenly fall apart. Despite the constant addition of food, sunlight, and medical care, entropy catches up, and when it does, it catches up fast. I haven't been able to find that chart, but it models the nonlinearity of the information entropy implicit in the human body.